The peculiar genetic background of each human being can affect individual drug response and modulate unwanted side effects. A single variation in a key gene involved in drug metabolism may influence drug efficacy, leading to potential severe side effects for particular groups of patients.
A variable drug response is a common scenario in neurodegenerative disorders, where each patient may also have specific disease development, a clinical finding that underscores the importance of taking into account the patient genetic background.
My general goal in the project PEGASO is to develop the first personalized body-on-chip platform to collect data useful for the selection of the most suitable genetic profile of patients to be enrolled in the clinical trials of new drugs, with the aim to improve efficacy, reduce side effects and the percentage of drug failure during the clinical phase, with a remarkable social and economic beneficial impact.
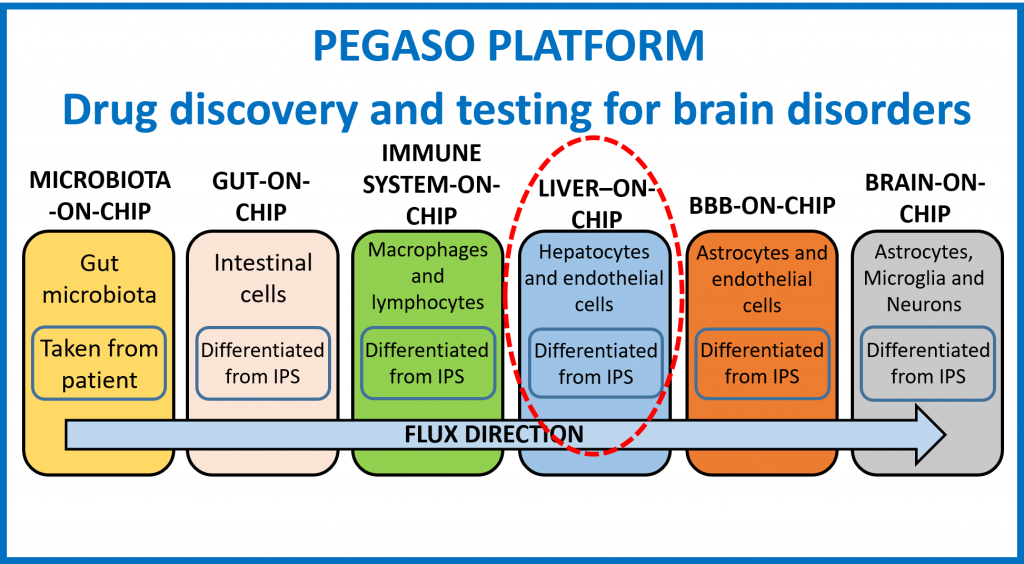
The platform relies on multiple sensorised, optically accessible organ-on-chip microfluidic devices, each modelling a specific body compartment, loaded with patient-specific induced pluripotent stem cell (IPSC) models, representing all the key organs/biological systems involved in drug metabolism and pharmacokinetics. The platform features a microbiota compartment to culture patient gut microbial cells to evaluate their contribution to drug metabolism and a liver-on-chip device to take into account the liver-mediated drug modification/activation pathways as occurs in vivo.
PEGASO methodology targets four goals: (a) selection of the most suitable protocols to differentiate and culture IPSC in all the organ-on-chip devices composing the platform; (b) adaption of some organ-on-chip prototypes obtained in my ERC project for PEGASO purposes; (c) development ex novo of the liver organ-on-chip device, not included in my ERC platform; (d) preliminary validation of the platform with Donepezil, a drug approved for Alzheimer’s disease, in two clinically relevant scenarios by selecting patients with and without genetic variations that can affect Donepezil pathway in vivo.
PEGASO has a ground-breaking potential: it might speed-up the current drug development protocol remarkably contributing to reducing the drug failure risk and potential collateral effects on patients enrolled in clinical trials, thanks to a reliable, genetically driven selection of the best responding patients.
PEGASO has a remarkable scientific impact: its platform represents an innovative powerful tool to investigate in vitro at the cellular level, in a multiorgan scenario, a reliable risks/benefit ratio of new tested drugs taking into account the individual profile of the patient.
PEGASO has a technological impact: it will result in the first body-on-chip platform representing up to six different biological systems that, thanks to its versatility, can be used in the personalisation of therapeutics for brain but also non brain-centred diseases.
MIUR FARE 2019 “PEGASO” (“PErsonalisation of induced pluripotent stem cell-based body-on-chips for in vitro testinG of therApeutic agentS against brain disOrders”; project code R18WWPCXLY)